GLP-1 Drugs: The Complete 2026 List
There are eight FDA-approved GLP-1 receptor agonists on the US market in 2026, plus compounded semaglutide and tirzepatide through telehealth. This page lists them all with indication, dosing, cost, and key trial outcomes.
Last updated May 1, 2026.
Licensed providers · All 50 states · No insurance required
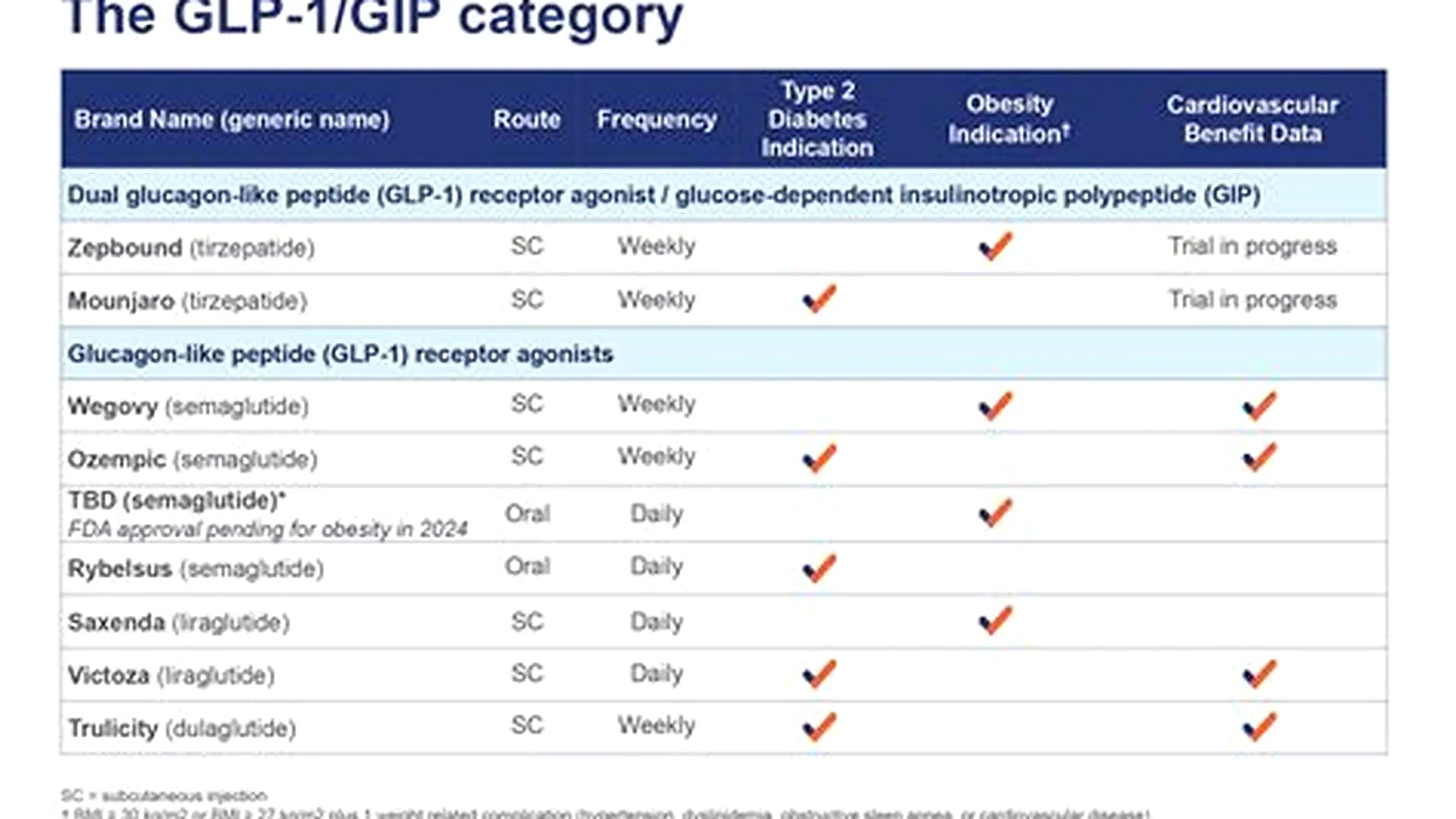
Quick Reference Table
| Drug | Molecule | Form | Approved for | Cash |
|---|---|---|---|---|
| Ozempic | Semaglutide | Weekly injection | Type 2 diabetes | $950–1,050 |
| Wegovy | Semaglutide | Weekly injection | Weight management (BMI 30+, or 27+ with comorbidity); Cardiovascular risk reduction | $1,300+ |
| Rybelsus | Semaglutide | Oral daily | Type 2 diabetes | $950+ |
| Mounjaro | Tirzepatide | Weekly injection | Type 2 diabetes | $1,070+ |
| Zepbound | Tirzepatide | Weekly injection | Weight management; Obstructive sleep apnea (in obesity) | $1,060+ |
| Saxenda | Liraglutide | Weekly injection | Weight management | $1,350+ |
| Victoza | Liraglutide | Weekly injection | Type 2 diabetes | $900+ |
| Trulicity | Dulaglutide | Weekly injection | Type 2 diabetes | $980+ |
Detailed Cards
Ozempic
injectionSemaglutide · Novo Nordisk
- Approved for: Type 2 diabetes
- Dosing: Weekly injection, 0.25–2.0 mg
- Cash price: $950–1,050/month
Wegovy
injectionSemaglutide · Novo Nordisk
- Approved for: Weight management (BMI 30+, or 27+ with comorbidity), Cardiovascular risk reduction
- Dosing: Weekly injection, 0.25–2.4 mg
- Cash price: $1,300+/month
Rybelsus
oralSemaglutide · Novo Nordisk
- Approved for: Type 2 diabetes
- Dosing: Daily oral tablet, 3/7/14 mg
- Cash price: $950+/month
Mounjaro
injectionTirzepatide · Eli Lilly
- Approved for: Type 2 diabetes
- Dosing: Weekly injection, 2.5–15 mg
- Cash price: $1,070+/month
Zepbound
injectionTirzepatide · Eli Lilly
- Approved for: Weight management, Obstructive sleep apnea (in obesity)
- Dosing: Weekly injection, 2.5–15 mg
- Cash price: $1,060+/month
Saxenda
injectionLiraglutide · Novo Nordisk
- Approved for: Weight management
- Dosing: Daily injection, 0.6–3.0 mg
- Cash price: $1,350+/month
Victoza
injectionLiraglutide · Novo Nordisk
- Approved for: Type 2 diabetes
- Dosing: Daily injection, 0.6–1.8 mg
- Cash price: $900+/month
Trulicity
injectionDulaglutide · Eli Lilly
- Approved for: Type 2 diabetes
- Dosing: Weekly injection, 0.75–4.5 mg
- Cash price: $980+/month
By Molecule: What Each Active Ingredient Does
Semaglutide
The most prescribed GLP-1 in the US. Brand names Ozempic (T2D), Wegovy (weight), Rybelsus (T2D, oral). Average weight loss ~15% in STEP-1. Full detail on semaglutide.
Tirzepatide
Dual GIP/GLP-1 agonist. Brand names Mounjaro (T2D), Zepbound (weight, OSA). Average weight loss ~22% at top dose in SURMOUNT-1. Full detail on tirzepatide.
Liraglutide
First once-daily injectable GLP-1. Brand names Victoza (T2D) and Saxenda (weight). Older, less potent than weekly options. Saxenda is FDA-approved for adolescents aged 12+.
Dulaglutide
Weekly injectable for type 2 diabetes (Trulicity). Cardiovascular benefit demonstrated in REWIND trial. Often well-tolerated.
Exenatide
First GLP-1 RA, approved 2005. Two formulations: twice-daily Byetta and weekly Bydureon. Largely replaced by newer drugs.
Lixisenatide
Daily injectable (Adlyxin). Limited use in the US; more common internationally.
FDA Approval Timeline
- 2005 — Exenatide (Byetta)
- 2010 — Liraglutide (Victoza)
- 2012 — Exenatide extended-release (Bydureon)
- 2014 — Albiglutide (Tanzeum; later discontinued)
- 2014 — Liraglutide 3 mg (Saxenda) for obesity
- 2014 — Dulaglutide (Trulicity)
- 2016 — Lixisenatide (Adlyxin)
- 2017 — Semaglutide injection (Ozempic)
- 2019 — Oral semaglutide (Rybelsus)
- 2021 — Semaglutide 2.4 mg (Wegovy) for obesity
- 2022 — Tirzepatide (Mounjaro) for T2D
- 2023 — Tirzepatide (Zepbound) for obesity
- 2024 — Zepbound expanded for OSA in obesity
By Indication
FDA-Approved for Weight Management
- Wegovy (semaglutide 2.4 mg)
- Zepbound (tirzepatide)
- Saxenda (liraglutide 3 mg)
FDA-Approved for Type 2 Diabetes
- Ozempic (semaglutide)
- Rybelsus (semaglutide oral)
- Mounjaro (tirzepatide)
- Trulicity (dulaglutide)
- Victoza (liraglutide)
- Byetta / Bydureon (exenatide)
- Adlyxin (lixisenatide)
Other FDA-Approved Indications
- Wegovy — Cardiovascular risk reduction in established CV disease
- Zepbound — Obstructive sleep apnea in obesity
- Ozempic — Cardiovascular risk reduction in T2D
- Trulicity — Cardiovascular risk reduction in T2D
- Saxenda — Weight management in adolescents (12+)
Pipeline: What's Coming Next
Several late-stage GLP-1 and combination drugs are in clinical trials in 2026:
- Retatrutide (Lilly) — triple agonist (GIP/GLP-1/glucagon). Phase 3 trials reported the largest weight loss yet (~24%).
- Orforglipron (Lilly) — oral small-molecule GLP-1 agonist, easier to manufacture and ship than peptides.
- CagriSema (Novo Nordisk) — combination of cagrilintide and semaglutide.
- Survodutide (Boehringer Ingelheim) — dual GLP-1/glucagon agonist.
How to Choose Between Them
Most patients have two main decisions:
- Semaglutide vs tirzepatide. Tirzepatide produces larger weight loss on average but is somewhat newer. Semaglutide has a longer safety record and more cardiovascular outcomes data.
- Brand vs compounded. Brand carries FDA manufacturing oversight; compounded is significantly cheaper. See compounded semaglutide and compounded tirzepatide.
A telehealth or in-person provider will help you weigh side-effect history, insurance coverage, cost preference, and goals.
Frequently Asked Questions
What are all the GLP-1 drugs available? +
Which GLP-1 is strongest? +
Which GLP-1 is cheapest? +
Which GLP-1 has fewest side effects? +
Related Reading
Sources
- FDA prescribing information for all listed drugs.
- STEP, SURMOUNT, SURPASS, SUSTAIN, REWIND, LEADER clinical trial publications.